22 November 2022: Clinical Research
Macronutrients Management for Growth in Neonates with Congenital Gastrointestinal Malformation
Timea E. Brandibur12AC, Aniko Maria Manea12AF, Abhinav SharmaDOI: 10.12659/MSM.938106
Med Sci Monit 2022; 28:e938106
Abstract
BACKGROUND: Congenital gastrointestinal (GI) malformations are developmental disorders that can result in secondary intestinal failure. Nutrient intakes must be adapted according to the newborn’s nutritional requirements based on frequent anthropometric and biochemical assessments. Deficiencies or excess of a macronutrient can hinder the growth of the newborn.
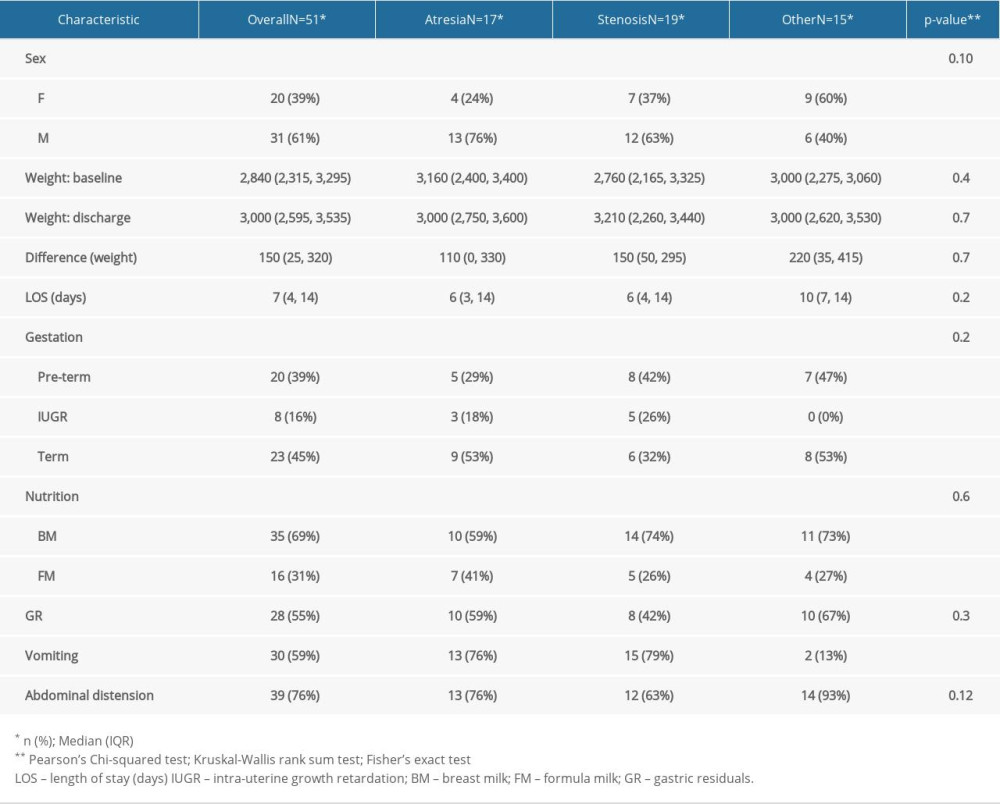
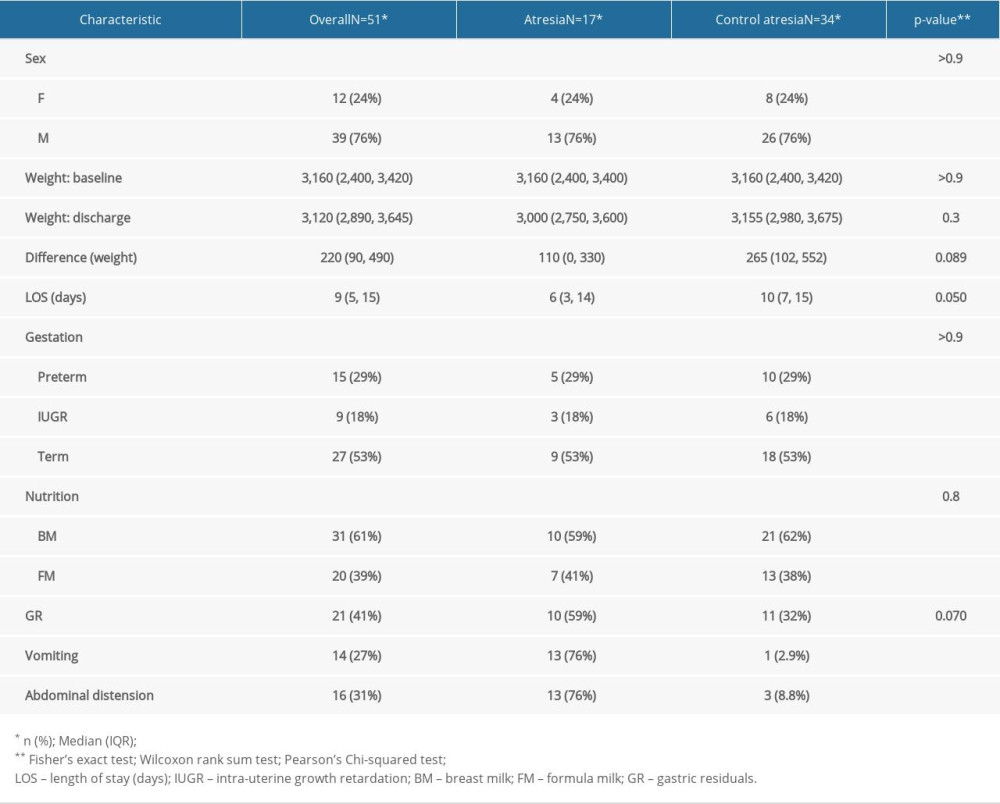
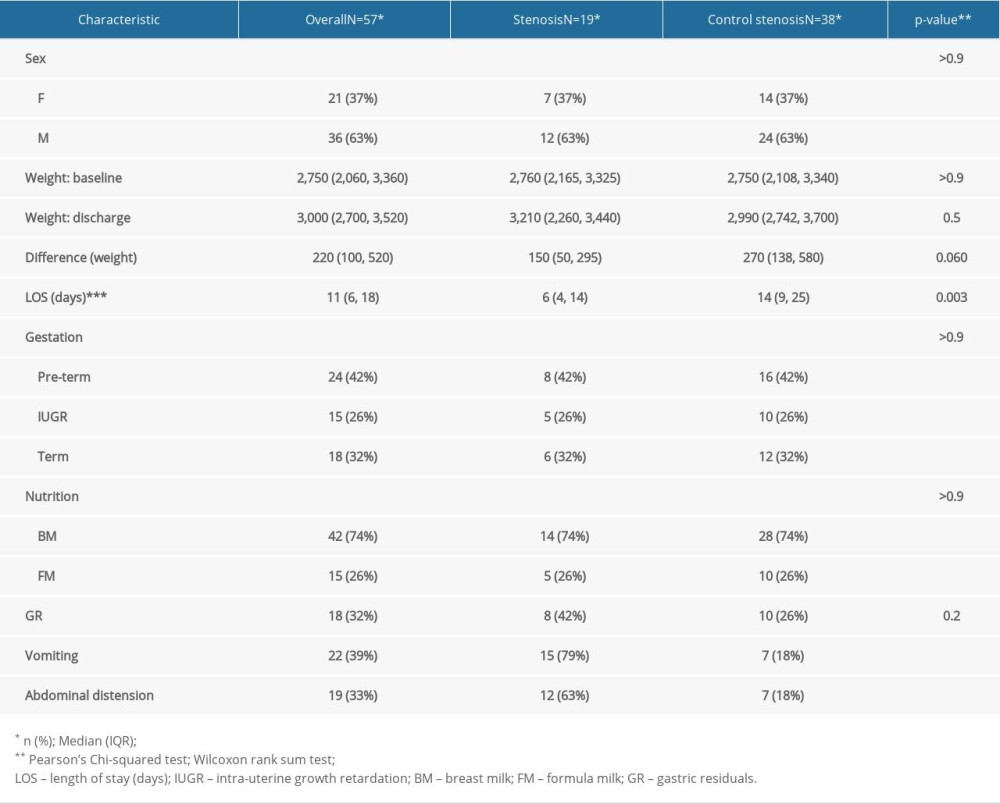
MATERIAL AND METHODS: To assess the clinical condition of newborns with GI malformations, together with the postoperative nutritional status of newborns who underwent surgery due to congenital GI malformations, we performed a case-control study. The study group comprised newborns with digestive malformations (n=51) and the control group consisted of newborns without digestive pathologies (n=102), matched by sex, gestational age, and weight at admission.
RESULTS: Bivariate comparisons and multiple logistic regression analyses were performed. A P value <0.05 was considered to be statistically significant; these were observed in abdominal distension, gastric residue, and vomiting. The duration of hospitalization was shorter in the case group, as on average, they were transferred to the Pediatric Surgery Department on the 6th day, where they further remained admitted to treat the underlying cause. Differences between groups in administration of breast milk versus formula were not statistically significant.
CONCLUSIONS: We concluded that the clinical examination had a major role in early detection of digestive malformations and in the effective management of specific necessary nutrition. Proper evaluation of when to start enteral feeding can help post-surgical cases to recover faster, minimizing complications. Further studies are required to assess how financial factors affect implementation of the standardized guidelines of nutrition in children and to find possible solutions to financial constraints.
Keywords: Digestive System Abnormalities, Enteral Nutrition, nutrients, Child, Female, Infant, Newborn, Humans, Case-Control Studies, Gastrointestinal tract, Gestational Age, Milk, Human
Background
Congenital gastrointestinal malformations are developmental disorders, or embryopathies, which can involve the bowels or other organ systems [1]. Intestinal failure is defined as a significant reduction in the functional gut mass, resulting in insufficient absorption of essential nutrients necessary to sustain life, including macronutrients (proteins, lipids, and carbohydrates, micronutrients, electrolytes, and most importantly, water [2–5]. Intestinal failure can occur due to surgical resection of the bowel, congenital anomalies, or functional/motility disorders. As such, these individuals require parenteral nutrition (PN) for survival. The subsequent nutritional, medical, and surgical aspects of care are complex [6,7]. The incidence is higher in premature infants with a gestational age of less than 37 weeks than in full-term infants [8]. Pediatric intestinal failure usually occurs secondary to other disorders, and short bowel syndrome (SBS) is a leading etiology [6]. It is characterized by a lack of absorptive capacity due to the severely reduced mucosal surface area. Necrotizing enterocolitis is the leading cause of neonatal SBS [9,10]; other etiologies include gastroschisis, which has a 30% increase in prevalence [11,12], intestinal atresia, malrotation with volvulus, and Hirschsprung’s disease [9].
The standard imaging study for gastrointestinal (GI) pathologies in infants is plain abdominal radiography together with conventional contrast studies. The management of congenital gastrointestinal malformations necessitates a multi-disciplinary approach that includes neonatologists, gynecologists, pediatric surgeons, gastroenterologists, and nurses. Parenteral nutrition (PN) is the life-saving therapy of choice in the management of intestinal failure. It involves a complex mix of appropriate macronutrients such as carbohydrates, proteins, lipids, and electrolytes, as well as micronutrients, delivered through an intravenous infusion to provide essential nutrition [11,13]. The main goal in therapy is to deliver the maximum tolerated quantity of enteral nutrition (EN), supported by PN [14]. The necessity of maintaining growth has to be balanced with the excess macronutrient intake by PN. For this purpose, strict monitoring of laboratory parameters is of extreme importance for newborns on long-term PN [15]. Therefore, it is preferred to initiate early enteral nutrition to prevent the adverse effects of parenteral nutrition. The rate of advancement of enteral feeding depends on stoma/stool output, vomiting, and abdominal distension [16]. The optimal enteral formula has not yet been established [17]. A large-scale, geographically comprehensive, multicenter, prospective cohort study was organized by N.J. Wright, comparing the different possibilities of managing and watchig the outcomes of a few common congenital problems in different countries having diffferent per capita income, results of which can be beneficial, taking into account how financial problems affect the health care systems and have immense negative impact on patient health and recovery [18].
Material and Methods
DETERMINATION OF GASTROINTESTINAL MALFORMATION:
All newborns were clinically examined daily by the neontologist and weighed by the nurse with the same scale. The type of milk and parenteral infusions administered were monitored daily in the observation sheet. The diagnosis of congenital abnormalities depended on the location of the abnormality along the GI tract. Abnormalities in the upper GI tract, the esophagus, the stomach, and the duodenum tend to rapidly become more problematic, opposed to those in the lower GI tract, such as the intestines, the rectum, and the anus, which take longer to become apparent.
STATISTICAL ANALYSIS:
Statistical analyses were performed and the group was compared using ANOVA. If ANOVA could not be applied, we used the Kruskal-Wallis rank sum test, Fisher’s exact test, or Pearson’s chi-squared test. A
Results
TOLERANCE OF EARLY ENTERAL NUTRITION:
During postoperative follow-up, abdominal distention was the most common observation, followed by repeated vomiting, in children who underwent surgery. Difficulties in nutritional recovery were encountered in 56.8% of cases (n=29). We also found a slow pace of nutritional recovery in all newborn cases. Poor weight recovery was present in 61.5% of the cases, 35% had malabsorption syndrome, 15% needed gastrostomy replacement, and surgery was performed in 10% of the cases. In addition to that, prolonged use of oro/nasogastric tubes accentuated pre-existing respiratory distress or produced a long-term aversion to oral food in 68.6% of the cases (n=35). Bodyweight decreased at a rate of 15% in newborns with stoma present in the first 10 days after hospitalization. The average length of hospital stay was longer in newborns who received long-term parenteral nutrition. Furthermore, early initiation of enteral feeding (18% of newborns) was achieved at the time of resumption of intestinal transit in premature infants with a gestational age greater than 32 weeks but lesser than the normal range of 38–42 weeks of gestation, and the number of days needed to reach full enteral nutrition was longer. Return to enteral feeding after parenteral nutrition in infants with gastric malformation was usually performed around 7–10 days postoperatively. Protein intake was 2.5 g/kg/day in full-term newborns and 4.5 g/kg/day for preterm very low birth weight (VLBW) and extremely low birth weight (ELBW) infants in both groups, while the lipid dose was increased to 4 g/kg/day in the postoperative period. Glucose can be synthesized from other sources by gluconeogenesis; therefore, there was no minimum required glucose from dietary sources. The milk used was 69% breast milk together with specific formulas for premature babies, whereas extensively hydrolyzed formulas were given for the full-term newborns. Patients were given 10 ml/kg/day of formula through a nasogastric tube, divided into 8 meals, and the feeding amount was increased by 10–15 ml/kg/day increment if it was adequately tolerated. Full feed was considered when the newborn tolerated 130 ml/kg/day. In the comparison between case and control groups, control subjects were more likely to have received higher enteral total fluid volume and calories, have reached full enteral feeds (defined as enteral feeds of at least 120 cc kg−1 per day with no use of parenteral nutrition), and to have fewer feeding interruptions than case patients.
Discussion
In the neonatal period, the nutritional requirements are higher compared to other pediatric groups due to the limited energy resources, pathology, and accentuated growth rate. The nutritional needs provided by enteral nutrition are adjusted according to weight and gestational age. The guidelines set by the American Society for Parenteral and Enteral Nutrition (ASPEN), the Society of Critical Care Medicine (SCCM), and the European Society of Pediatric and Neonatal Intensive Care should be carefully followed for a better and faster recovery of patients [19–21]. However, due to limited resources, it is sometimes difficult to implement such guidelines, as happened in our study. The purpose of EN in the critical newborn is to ensure proper growth, development, and prophylaxis of complications related to nutrition. Moreover, finding a balance of appropriate nutritional intake to meet metabolic demands is frequently difficult during the perioperative period [22]. Enteral nutrition can be started early in the postoperative period in neonates [23]. During enteral nutrition, some complications can occur, such as mechanical or digestive complications leading to the interruption of enteral nutrition; these can manifest as bilious vomiting, abdominal distension, and intestinal perforation [23–25]. Nevertheless, early postoperative enteral feeding did not increase the incidence of postoperative gastrointestinal complications [7,26,27]. Gastric residue should also be checked before each feeding [28]. The rate of advancement of enteral feedings should be individualized, with careful monitoring of stool/stoma output, vomiting, and abdominal distention. Continuous enteral feeding allows complete saturation of molecules in intestinal transporters and increased absorption of lipids, proteins, calcium, zinc, and copper [29]. EN with human milk is also associated with higher survival and is associated with shorter hospital stay without major complications [30]. Studies on the metabolic response in infants who undergo surgery indicate that protein turnover is influenced by fat-to-carbohydrate ratio; thus, parenteral nutrition support with amino acids rather than dextrose is useful to control this state [31,32]. Furthermore, extensively or partially hydrolyzed formulas have greater digestibility with lower energy consumption of protein hydrolysates compared to whole proteins [33,34]. Recognizing the best type of formula milk to use requires extensive research; the use of Extensively Hydrolyzed Formula is justified by the increased risk of protein allergy characterizing newborns after intestinal surgery, whereas macronutrients in a complex form were found to better promote bowel adaptation. On the other hand, the use of the semi-elemental or elemental formula is suggested in those patients who have an intolerance to conventional preterm/term formulas. Our study results suggest that children should be administered enteral feed as soon as possible to avoid prolonged use of parenteral nutrition, but at the same time the tolerance to enteral feedings should be evaluated. Early diagnosis of gastrointestinal pathologies or malformations can avoid unnecessary delay in treatment and provide nutritional care to the newborns. A number of national and international guidelines are used in many countries, but, unfortunately, due to financial constraints we are not able to follow these completely. Government-funded hospitals have limitations and milk formulas provided are ever-changing depending on the budget and vendors approved, which makes it impossible for doctors to prescribe feedings meeting international norms.
Conclusions
The ideal enteral nutrition formula for neonates with gastric malformation remains controversial. Breast milk and commercially available elemental formulas are associated with a reduced duration of PN dependence. Early enteral feeding can increase survival, accelerate recovery of gastrointestinal function, and improve the nutritional status of newborns. The time of onset of enteral nutrition depends on several factors, such as gestational age, intestinal functional status, milk formula used (polymeric vs elemental), and route of administration (stomach vs jejunum). The management of a newborn with a gastrointestinal malformation necessitates qualified care for food administration in particular; slow nutritional recovery should be expected together with frequent complications. Neonatal digestive pathology remains a major cause of mortality and morbidity. Neonates should be evaluated thoroughly on a case-by-case basis to evaluate the need for parenteral nutrition and when to decide when to switch to enteral feeding. Neonates undergoing major surgeries need more attention and support; not only for healing, but also to achieve optimal growth and development. However, more studies are required to discover the optimal feeding formula and routine clinical therapy for neonates with GI malformations.
References
1. Fotis L, Burns AJ, Thapar N: Gastrointestinal tract: Congenital abnormalities https://onlinelibrary.wiley.com/doi/10.1002/9780470015902.a0002138.pub2
2. Goulet O, Ruemmele F, Causes and management of intestinal failure in children: Gastroenterology, 2006; 130(2 Suppl 1); S16-28
3. Gutierrez IM, Kang KH, Jaksic T, Neonatal short bowel syndrome: Semin Fetal Neonatal Med, 2011; 16(3); 157-63
4. Allan P, Lal S, Intestinal failure: A review: F1000Res, 2018; 7; 85
5. Nicolescu CM, Osakwe H, Pop A, Nicolescu LC, Evaluation of plasma albumin as a potential prognostic biomarker in patients with traumatic SIRS: Rev Chim, 2017; 68(9); 2181-88
6. Mangalat N, Teckman J, Pediatric intestinal failure review: Children (Basel), 2018; 5(7); 100
7. Osakwe HI, Dragomir C, Nicolescu C, Boia ES, The challenges of managing and following-up a case of short bowel in eastern europe: Int J Surg Case Rep, 2016; 26; 187-92
8. Wales PW, Christison-Lagay ER, Short bowel syndrome: epidemiology and etiology: Semin Pediatr Surg, 2010; 19(1); 3-9
9. Amin SC, Pappas C, Iyengar H, Maheshwari A, Short bowel syndrome in the NICU: Clin Perinatol, 2013; 40(1); 53-68
10. Neu J, Walker WA, Necrotizing enterocolitis: N Engl J Med, 2011; 364(3); 255-64
11. Duggan CP, Jaksic T, Pediatric intestinal failure: N Engl J Med, 2017; 377(7); 666-75
12. Goulet O, Finkel Y, Kolaček S, Puntis J, Chapter 5.2.1. Short bowel syndrome: Half a century of progress: J Pediatr Gastroenterol Nutr, 2018; 66(Suppl 1); S71-S76
13. Wilmore DW, Groff DB, Bishop HC, Dudrick SJ, Total parenteral nutrition in infants with catastrophic gastrointestinal anomalies: J Pediatr Surg, 1969; 4(2); 181-89
14. Diamanti A, Calvitti G, Martinelli D, Etiology and management of pediatric intestinal failure: Focus on the non-digestive causes: Nutrients, 2021; 13(3); 786
15. Wilmore DW, Dudrick SJ, Growth and development of an infant receiving all nutrients exclusively by vein: JAMA, 1968; 203(10); 860-64
16. Wessel JJ, Kocoshis SA, Nutritional management of infants with short bowel syndrome: Semin Perinatol, 2007; 31(2); 104-11
17. Rudolph JA, Squires R, Current concepts in the medical management of pediatric intestinal failure: Curr Opin Organ Transplant, 2010; 15(3); 324-29
18. Wright NJGlobal PaedSurg Research Collaboration, Management and outcomes of gastrointestinal congenital anomalies in low, middle and high income countries: Protocol for a multicentre, international, prospective cohort study: BMJ Open, 2019; 9(8); e030452
19. https//www.nutritioncare.org/Guidelines_and_Clinical_Resources/Enteral_Nutrition_Resources/
20. Tume LN, Valla FV, Joosten K, Nutritional support for children during critical illness: European Society of Pediatric and Neonatal Intensive Care (ESPNIC) metabolism, endocrine and nutrition section position statement and clinical recommendations: Intensive Care Med, 2020; 46(3); 411-25
21. Mehta NM, Skillman HE, Irving SY, Guidelines for the provision and assessment of nutrition support therapy in the pediatric critically ill patient: Society of Critical Care Medicine and American Society for Parenteral and Enteral Nutrition: Pediatr Crit Care Med, 2017; 18(7); 675-715
22. Khlevner J, Antino J, Panesar R, Chawla A, Establishing early enteral nutrition with the use of self-advancing postpyloric feeding tube in critically ill children: J Parenter Enteral Nutr, 2012; 36(6); 750-52
23. Berger D, Roulet M, Early postoperative enteral feeding through a needle catheter jejunostomy: Z Kinderchir, 1984; 39(5); 328-31
24. Suri S, Eradi B, Chowdhary SK, Early postoperative feeding and outcome in neonates: Nutrition, 2002; 18(5); 380-82
25. Miranda da Silva Alves F, Miranda ME, Nutritional management and postoperative prognosis of newborns submitted to primary surgical repair of gastroschisis: J Pediatr (Rio J), 2016; 92(3); 268-75
26. Agladze NDThe assessment of risk factors of the atresia of the gastro-intestinal tract: Georgian Med News, 2006; 137; 71-74
27. DeLorimier AA, Fonkalsrud EW, Hays DM, Congenital atresia and stenosis of the jejunum and ileum: Surgery, 1969; 65(5); 819-27
28. Btaiche IF, Chan LN, Pleva M, Kraft MD, Critical illness, gastrointestinal complications, and medication therapy during enteral feeding in critically ill adult patients: Nutr Clin Pract, 2010; 25(1); 32-49
29. Harris JB, Neonatal short bowel syndrome: Newborn and Infant Nursing Reviews, 2007; 7(3); 131-42
30. Gulack BC, Laughon MM, Clark RH, Enteral feeding with human milk decreases time to discharge in infants following gastroschisis repair: J Pediatr, 2016; 170; 85-89
31. Pierro A, Metabolism and nutritional support in the surgical neonate: J Pediatr Surg, 2002; 37(6); 811-22
32. Donatelli F, Schricker T, Mistraletti G, Postoperative infusion of amino acids induces a positive protein balance independently of the type of analgesia used: Anesthesiology, 2006; 105(2); 253-59
33. Szajewska H, Horvath A, Meta-analysis of the evidence for a partially hydrolyzed 100% whey formula for the prevention of allergic diseases: Curr Med Res Opin, 2010; 26(2); 423-37
34. Thompkinson DK, Kharb S, Aspects of infant food formulation: Comprehensive Reviews in Food Science and Food Safety, 2007; 6(4); 79-102
In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952